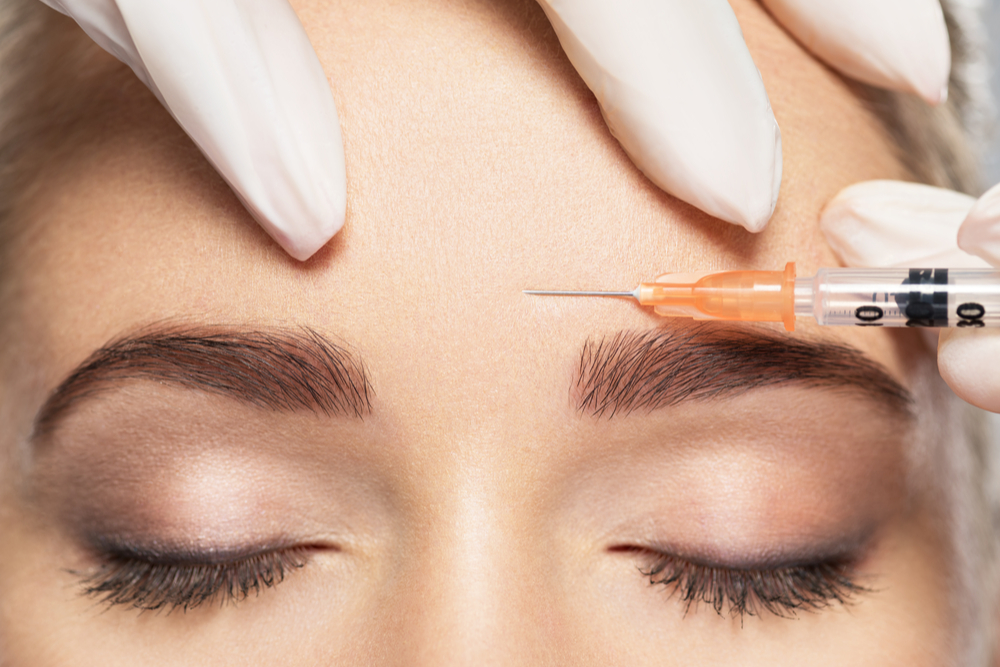
- Xeomin is a botulinum toxin that has a variety of aesthetic and medical applications.
- There’s little to no downtime involved with receiving a Xeomin injection
- Xeomin is generally considered to be very safe, however there are some side effects to be cognizant of
- Although similar, there are some differences between Xeomin and Botox
Botox is the name that probably comes to mind when you think of treating facial wrinkles, but you might be surprised to learn that there are several botulinum toxin alternatives available, one of which is Xeomin.
What Is Xeomin?
A relative newcomer to the U.S. market, Xeomin is an injectable filler used to treat frown lines. It incorporates the same active ingredient as Botox, botulinum toxin type A, except Xeomin is a slightly purer form of the toxin, which is believed to be beneficial to long-term patients in particular.
Xeomin is manufactured by the multinational healthcare company Merz Pharma GmbH & Co. In 2011, the drug gained FDA approval for cosmetic use in the United States, although it has been available in Europe under the brand name Bocouture since 2008.
What Is Xeomin Used to Treat?
Xeomin has a variety of aesthetic and medical applications.
In July 2011, Xeomin gained FDA approval for use in the temporary improvement of moderate to severe glabellar lines (also known as frown lines).
Botox also has FDA approval for treating crow’s feet and forehead lines.
Beyond cosmetic applications, Xeomin has also been approved by the FDA to treat a variety of medical conditions, including:
- Cervical dystonia
- Blepharospasm
- Adult upper limb spasticity
- Sialorrhea (excessive drooling)
What Is Involved with the Xeomin Procedure?
During treatment, your doctor will use a very thin needle to inject Xeomin into the forehead muscles responsible for the frown lines between your eyebrows.
You may feel a slight pinch, but the procedure typically isn’t considered to be painful. Your doctor may use a topical anesthetic prior to treatment to alleviate discomfort.
The treatment usually takes less than 30 minutes from start to finish.
Recovery Time After Receiving a Xeomin Injection
As with most botulinum toxin treatments, there’s little to no downtime after receiving a Xeomin injection, with most people returning to their regular activities immediately afterward.
You may experience some slight bruising, swelling and redness near the injection site. These side effects tend to resolve within a couple of days, and will have usually already started to fade within a few hours of treatment.
Your doctor may recommend avoiding strenuous exercise for a few days following your Xeomin treatment, as the increased blood pressure from physical activity can potentially exacerbate bruising.
You might also be asked to refrain from rubbing or applying pressure to your face for a few hours to reduce the risk of Xeomin affecting any areas of the body outside of the injection site. Your doctor may also advise the use of Arnica cream to manage swelling.
In order to get the best results and minimize the risk of potential complications occurring, it’s important that you follow your doctor’s instructions exactly as directed.
How Long Do the Effects of Xeomin Last?
Results are not immediately visible following the procedure, however, most people start to notice improvements within a week, and the full effects roughly one month later.
In research published in The Journal of Clinical and Aesthetic Dermatology, 98.1% of subjects reported improvement in their frown lines 28 days after treatment, while 85.6% reported improvements 84 days after treatment.
There is no tangible difference in longevity between Xeomin and Botox. Both last for approximately four months, with some variation between patients. Once Xeomin’s effects begin to wear off, you can return to your provider for another injection if you wish to maintain results.
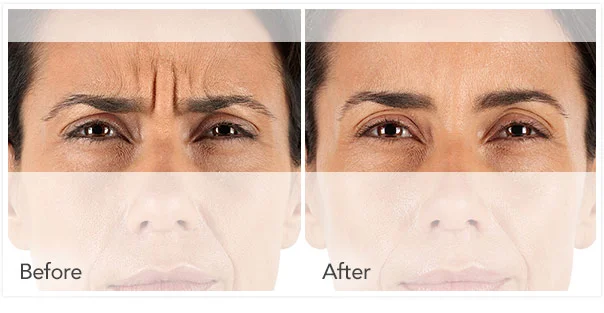
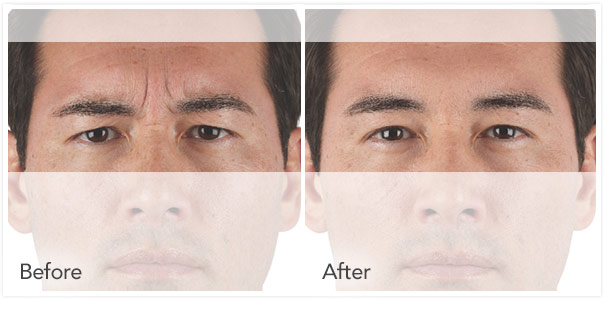
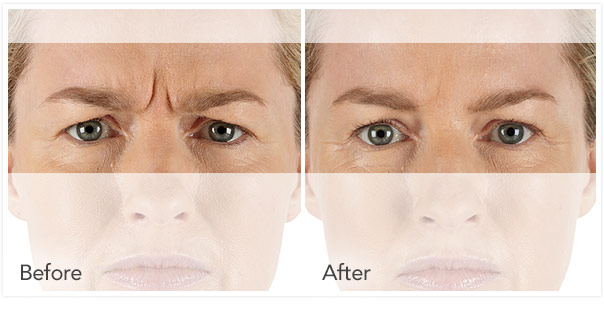
Xeomin Before and After Pictures
Xeomin Side Effects
Xeomin is generally considered to be very safe. Nevertheless, as with just about all cosmetic treatments, there are some potential side effects to be aware of.
Headaches are the most common side effect, but you could also experience:
- Dry eyes
- Rashes
- Dizziness
- Double vision
- Dry mouth
- Drooping eyelids
- Puffy eyes
- Reduced blinking
- Neck pain
In addition, as with other botulinum toxin injections, there is a risk that Xeomin could migrate away from the injection site and affect other parts of the body. If you experience any side effects that bother you or do not subside, you should seek medical advice immediately.
Treatment during pregnancy
There have been no studies on the effects of Xeomin during pregnancy. In light of this pregnant and nursing mothers should avoid Xeomin.
Allergies
It’s possible some people will have an allergic reaction to Xeomin. Should you experience any trouble breathing, lightheadedness, muscle weakness, or swelling of the face, lips, tongue or throat, contact your doctor right away.
How Much Does Xeomin Cost?
In the United States Xeomin costs approximately $10 per unit.
The number of units required for your treatment can vary depending on your unique anatomy and the injection site, so consider the costs below as only a rough estimate:
| Injection site | Units required | Price |
|---|---|---|
| Forehead | 10-15 | $100-$150 |
| Crow’s feet | 15-25 | $150-$250 |
| Glabella lines | 20 | $200 |
Xeomin is slightly less expensive than Botox, which typically costs approximately $12 per unit.
It’s important to remember that costs can vary quite significantly depending on your geographical location and the fees set by your provider.
The prices outlined above don’t take into account other costs associated with the Xeomin procedure, so the total cost of your treatment may be higher.
Xeomin vs Botox: What’s the Difference?
The main difference between Xeomin and Botox is that Xeomin uses a proprietary purification process to remove accessory proteins from botulinum toxin type A, creating a slightly different type of toxin known as incobotulinumtoxinA.
Why is this important?
It is believed that long-term exposure to the proteins found in Botox increase the chances of the body developing a resistance to botulinum toxin, making future treatments ineffective.
According to research published in Clinical, Cosmetic and Investigational Dermatology, a small percentage of long-term patients develop antibodies to the botulinum toxin molecule. In theory, Xeomin may reduce the risk of antibody formation, although this has yet to be proven in clinical studies.
- Fewer complexing proteins: As noted, the main difference between Xeomin and Botox is that Xeomin doesn’t contain as many complexing proteins as Botox. This means a patient can theoretically use Xeomin for long-term treatment without worrying about building a resistance to the botulinum toxin.
- Cost: Xeomin is slightly cheaper than Botox, making it a potentially better option for those on a budget.
- Easier distribution: Unlike Botox, Xeomin does not require refrigeration. This makes things a bit easier for providers, but has little impact on patients.
See this guide for more information on the differences between Xeomin and Botox.
Takeaway
It might not be as well-known as other injectable products, but Xeomin is proving to be an effective option for temporarily reducing the appearance of facial wrinkles. Xeomin’s primary difference lies in its special purification process, which results in a purer form of the product’s active ingredient, botulinum toxin. This makes it a good option for long-term patients, as there is less risk of the body developing a resistance to the product.
Find a Xeomin Provider near You
With its competitive prices and pure botulinum toxin formulation, Xeomin is an excellent option for anyone seeking an effective BOTOX alternative. To ensure the best possible results, it is important to Use the services of our medical review team .